A diagnosis of truncus arteriosus is one of the most serious things a parent can hear about their newborn. The words are unfamiliar, surgery is discussed within days, and the fear can feel overwhelming. What families need most at that point is clear information from someone with genuine expertise in complex congenital heart disease.
This page explains what truncus arteriosus is, why surgery is needed and what it involves, what recovery looks like, and what you can realistically expect for your child’s future. Dr. Alessandro Giardini has cared for children with complex congenital heart conditions including truncus arteriosus throughout his career at Great Ormond Street Hospital, and this guide reflects the kind of explanation he offers families in clinic.
What Is Truncus Arteriosus?
Truncus arteriosus, also called common arterial trunk, is a congenital heart defect present from birth. In a normal heart, two separate blood vessels leave the heart: the pulmonary artery, which carries oxygen-poor blood from the right side to the lungs, and the aorta, which carries oxygen-rich blood from the left side to the rest of the body. Both vessels develop from a single shared structure during fetal development, dividing into two at around five to eight weeks of pregnancy.
In truncus arteriosus, this division does not happen. A single large blood vessel (the truncus) leaves the heart, served by one valve (the truncal valve). Blood from both sides of the heart mixes inside this single vessel, and a mixture of oxygenated and deoxygenated blood travels to both the lungs and the body. This is inefficient and places enormous strain on the heart and lungs.
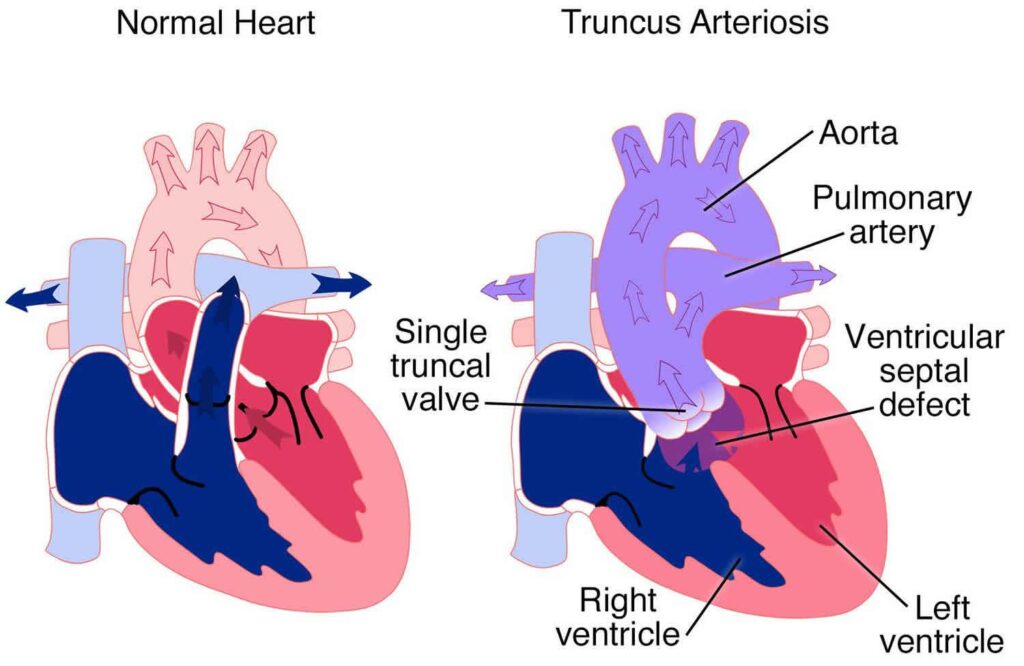
Almost all children with truncus arteriosus also have a large hole between the two lower chambers of the heart, called a ventricular septal defect (VSD). This is not a separate problem but part of the same developmental anomaly.
Truncus arteriosus accounts for approximately one per cent of all congenital heart defects and affects roughly one in every 10,000 to 14,000 births. It is rare but well understood, and surgical outcomes have improved substantially over recent decades.
Types of Truncus Arteriosus
Surgeons and cardiologists classify truncus arteriosus according to how the pulmonary arteries branch from the single trunk. In Type 1 (the most common), a short main pulmonary artery arises from the trunk and then divides into right and left pulmonary arteries. In Type 2, the right and left pulmonary arteries arise separately from the back of the trunk, close together. In Type 3, the pulmonary arteries arise from the sides of the trunk.
The type affects surgical planning but does not change the fundamental principles of treatment. The truncal valve itself may also be abnormal: it can have two, three, four, or even five leaflets, and significant leakage or narrowing adds complexity to management.
What Causes Truncus Arteriosus?
Truncus arteriosus develops because of abnormal fetal heart formation during early pregnancy. In most cases the exact trigger is unknown, and parents should understand clearly that this condition is not caused by anything they did or did not do during pregnancy.
Approximately 30 to 35 per cent of children with truncus arteriosus have 22q11.2 deletion syndrome, which includes DiGeorge syndrome. This chromosomal deletion affects development of the heart, immune system, parathyroid glands, and other systems. Children with 22q11.2 deletion need assessment of these broader health aspects alongside their cardiac care. Genetic testing forms part of the standard workup, and families benefit from access to clinical genetics advice.
In the remaining majority of cases, no specific genetic cause is found. Genetic testing after diagnosis gives families clarity and helps guide long-term planning for the child and for future pregnancies.
What Are the Symptoms of Truncus Arteriosus?
Many babies appear relatively well at birth, because the resistance in the lung blood vessels remains high in the first hours of life. As this resistance falls naturally over the first days and weeks, increasing blood floods into the lungs. This worsens both heart failure and oxygen delivery simultaneously.
Typical symptoms develop within the first days to weeks of life and include fast or laboured breathing, difficulty feeding and tiring easily during feeds, poor weight gain, sweating during feeding (a sign of increased cardiac effort), and a bluish tinge around the mouth, lips, and fingertips (cyanosis). A heart murmur is almost always present and audible on examination. Some babies have strongly throbbing pulses, reflecting the large run-off of blood from the truncus into the pulmonary arteries.
Without surgical treatment, truncus arteriosus is fatal, usually within the first year of life. Pulmonary hypertension (high blood pressure in the lung vessels) develops rapidly and becomes irreversible without early surgical correction. Prompt diagnosis and referral to a specialist cardiac centre are critical.
How Is Truncus Arteriosus Diagnosed?
Before Birth
Many cases receive a diagnosis before birth, either on the routine 20-week anomaly scan or on a fetal echocardiogram arranged because of a suspected cardiac abnormality. Antenatal diagnosis allows specialist counselling before birth, delivery planning at or near a cardiac centre, and prompt intervention without delay.
After Birth
When diagnosis occurs after birth, the combination of cyanosis, respiratory distress, and feeding difficulty prompts urgent investigation. The key tests include:
An echocardiogram (heart ultrasound) confirms the diagnosis, defines the anatomy of the truncus and its branches, assesses the truncal valve, identifies the VSD, and evaluates cardiac function. This is the single most important investigation and provides the information needed for surgical planning.
A chest X-ray typically shows an enlarged heart and increased blood vessel markings in the lungs, reflecting high pulmonary blood flow.
An ECG (electrocardiogram) assesses the electrical activity of the heart and identifies any rhythm abnormalities.
Pulse oximetry forms part of routine newborn screening in UK hospitals. A persistently low oxygen reading triggers further cardiac assessment.
Cardiac MRI or CT angiography may provide additional anatomical detail before surgery in complex variants.
Genetic testing, including chromosomal microarray analysis to detect 22q11.2 deletion, forms part of the standard assessment.
What Does Treatment Involve?
Surgery is the only treatment for truncus arteriosus. Without it, the condition is fatal. With it, most children survive and go on to live active lives.
Before Surgery
Babies need stabilisation before the operation. Medications to manage heart failure, including diuretics to reduce fluid overload, are started promptly. In some babies, additional respiratory support is needed.
Timing of Surgery
Surgery typically takes place within the first four to eight weeks of life, and in many centres within the first two to four weeks. Earlier surgery, before pulmonary hypertension becomes established, produces better outcomes.
What the Operation Involves
The surgical repair addresses three things at once. The VSD closes with a patch, directing all left ventricular blood through the truncal valve into what now functions as the aorta. The pulmonary arteries then separate from the truncus. Finally, a conduit (a tube containing a valve) connects the right ventricle to the pulmonary arteries, recreating the pathway for blood to reach the lungs. The surgeon may also repair the truncal valve if it leaks significantly.
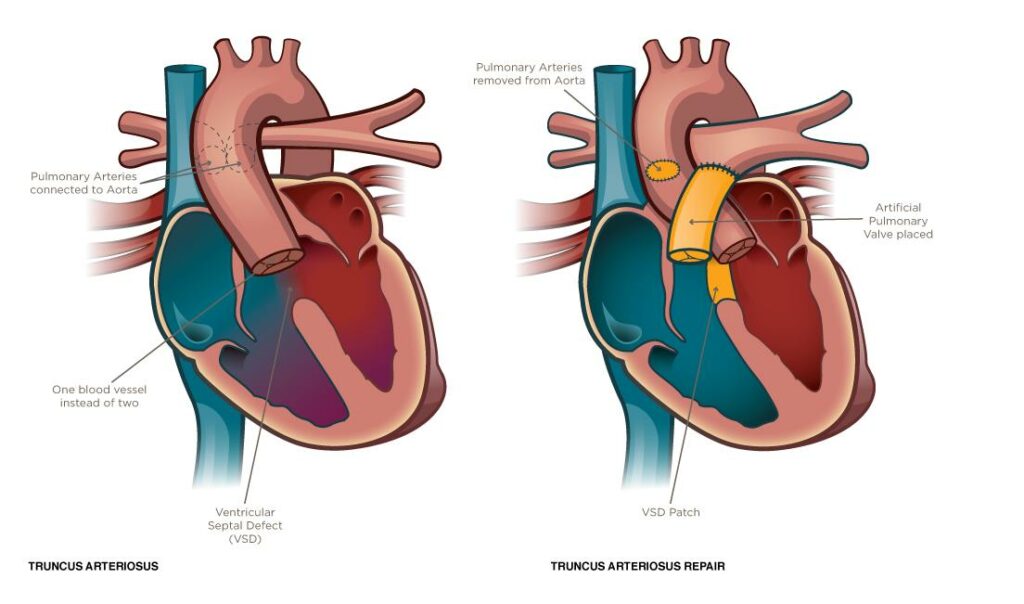
The operation takes place on cardiopulmonary bypass (the heart-lung machine), which temporarily takes over the function of the heart and lungs. It is complex surgery, performed at specialist congenital heart centres. Families receive detailed pre-operative explanation of the specific approach planned for their child.
Recovery After Surgery
Most babies spend one to three weeks in the cardiac intensive care unit following surgery, then move to the cardiac ward before going home. The surgery changes the pressures inside the heart, and the heart needs time to adapt. Possible issues during recovery include temporary rhythm disturbances requiring medication, fluid management challenges, and wound or chest infections. Babies with 22q11.2 deletion may have immune difficulties that require additional attention. Most children are well and gaining weight within a few weeks of surgery.
Will My Child Need Further Surgery?
This is one of the questions parents ask most often, and it deserves a direct answer: yes, further intervention is very likely at some point.
The conduit placed during the initial repair does not grow with the child. As the child grows, the conduit becomes relatively smaller. Over time, flow through it becomes restricted (conduit stenosis) or the valve inside begins to leak (conduit regurgitation). Both problems reduce cardiac efficiency and need correction.
Regular echocardiographic follow-up detects these changes before they become clinically urgent. When intervention is needed, two main options exist.
Open surgical conduit replacement has been the traditional approach and remains the standard in many centres and situations. Transcatheter pulmonary valve replacement (TPVR), using devices such as the Melody valve or Edwards Sapien valve, now offers a catheter-based alternative that avoids open chest surgery in suitable children. Interventional cardiologists deliver the new valve inside the existing conduit through a tube inserted into a blood vessel, substantially reducing recovery time. Not all children qualify for this approach, and the decision depends on the size and anatomy of the conduit, but TPVR represents a significant advance in managing truncus arteriosus over a lifetime.
The truncal valve (now functioning as the aortic valve) may also develop problems over time, particularly leakage. Significant truncal valve regurgitation may eventually need surgical repair or replacement. Rhythm problems (arrhythmias) can also develop in some children and adults with repaired truncus arteriosus, and monitoring for these forms part of long-term follow-up.
What Is the Long-Term Outlook?
The outlook after surgical repair of truncus arteriosus has improved dramatically over the past three decades. Contemporary surgical series report ten-year survival rates of 90 to 95 per cent for children who receive timely repair.
Most children who have successful repair grow normally, attend school, and participate in physical activities. Many reach adulthood and lead full, independent lives. The condition requires ongoing specialist follow-up throughout life, but this monitoring catches changes early and allows management at the right time.
Families should understand that truncus arteriosus is a lifelong condition in the management sense, not in the limiting sense. With the right care, most children do far better than parents fear in those frightening first days after diagnosis.
As part of normal care, children with repaired truncus arteriosus may need antibiotic prophylaxis before certain dental and surgical procedures, to reduce the risk of infective endocarditis (infection of the heart valves). Their cardiac team provides specific guidance on this.
Transition to Adult Care
As children with truncus arteriosus reach adolescence and early adulthood, their care transfers to an adult congenital heart disease (ACHD) service. This is a planned, supported process, not an abrupt handover. Adult cardiologists specialising in congenital heart disease continue the same programme of monitoring, managing conduit function, watching for arrhythmias, and assessing truncal valve status.
Young adults with truncus arteriosus can work, exercise, and in most cases plan pregnancies, though pregnancy in women with complex congenital heart disease needs specialist pre-conception counselling and careful monitoring throughout.
Dr. Giardini’s role at Great Ormond Street Hospital spans both the acute and long-term management of children with complex congenital conditions, and he has extensive experience guiding families through each transition point in their child’s care.
When Should You Seek Specialist Advice?
If your baby has received a diagnosis of truncus arteriosus, the urgent priority is specialist paediatric cardiology and cardiac surgery input at a centre experienced in complex congenital heart disease.
Frequently Asked Questions
Is truncus arteriosus life-threatening?
Yes, without treatment it is. Untreated, it causes progressive heart failure and pulmonary hypertension, and without surgery most babies do not survive the first year. With timely surgical repair, however, outcomes are very good and most children do well.
When is surgery usually done?
Surgery typically takes place within the first two to eight weeks of life. Earlier repair, before pulmonary hypertension becomes established, produces better outcomes. The exact timing depends on the baby’s condition and anatomy, and the specialist surgical team makes this decision.
Can truncus arteriosus be detected during pregnancy?
Yes, often. Clinicians identify many cases on the routine 20-week anomaly scan or on a fetal echocardiogram arranged following a suspicious finding. Antenatal diagnosis allows delivery planning at a specialist centre and prompt treatment after birth.
Will my child need more than one operation?
Almost certainly yes, over the course of their life. The conduit placed during the initial repair does not grow with the child and will need revision, either through open surgery or (in suitable cases) a catheter-based procedure such as transcatheter pulmonary valve replacement (Melody or Sapien valve). The truncal valve may also need attention over time.
Can my child live a normal life after surgery?
Most children do very well after successful repair and lead active, full lives. They attend school normally, participate in sport, and reach adulthood. Lifelong cardiac follow-up is needed, but for most families this becomes a routine part of life rather than a limiting one.
Is truncus arteriosus genetic?
It can be. Around 30 to 35 per cent of children with truncus arteriosus have 22q11.2 deletion syndrome (which includes DiGeorge syndrome). Genetic testing forms part of the standard assessment after diagnosis. Even when no genetic cause is found, families planning future pregnancies benefit from genetic counselling.
What are the warning signs in a newborn?
The main signs in the first days and weeks of life are fast or laboured breathing, difficulty feeding, poor weight gain, sweating during feeds, unusual tiredness, and a bluish colour of the lips or fingertips. Any baby showing these signs needs urgent medical assessment.
Does my child need lifelong follow-up?
Yes. Children and adults with repaired truncus arteriosus need regular echocardiographic monitoring, arrhythmia surveillance, and assessment of conduit and truncal valve function throughout life. This ongoing care is what allows problems to be detected and managed early, before they become clinically significant.
What is the conduit and why does it need replacing?
The conduit is the tube (usually containing a valve) that surgeons place to connect the right ventricle to the pulmonary arteries during the initial repair. Because it does not grow with the child, it becomes relatively smaller over time. When it narrows enough to restrict blood flow or the valve inside leaks significantly, it needs revision. In suitable children, a catheter-based procedure (transcatheter pulmonary valve replacement) can replace the valve without open surgery.
Should I seek a second opinion?
Many parents find a second opinion enormously valuable, particularly when planning surgery, facing conduit revision, or wanting independent assessment of their child’s long-term management plan. A second opinion does not delay care and often gives families greater confidence and clarity. Dr. Giardini welcomes referrals and self-referrals from families in this situation and offers Private Paediatric Cardiology consultations at his London clinics.
Author: Dr. Alessandro Giardini, MD, PhD
Written 09/04/2026

